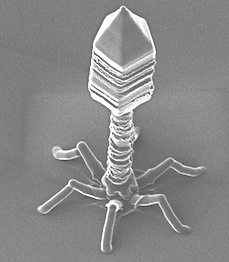
The Lancet ID, a monthly infectious diseases journal, renowned for the publication of high-quality peer-reviewed reviews and analysis from around the world, published in November, 2015, a publication about PhagoBurn project, called “Phage therapy for severe infections tested in the first multicentre trial” and written by Clare Sansom.